

IBT specializes in delivering top-notch reagents tailored for research and development purposes.
The global threat posed by flavivirus strains—including Dengue, Zika, West Nile Virus, and Yellow Fever—is no longer a distant concern for tropical regions. With climate change expanding the reach of mosquito vectors, the race for antiviral therapeutics and vaccine development has reached a critical financial and clinical juncture.
For scientists in infectious disease research, understanding the “commercial wall” is as vital as understanding the viral genome. While billions are invested annually, the path to regulatory submission remains one of the most challenging in modern medicine.
Despite decades of research, the cupboard for FDA-approved flavivirus therapeutics remains surprisingly bare.
To bridge the gap between a promising lead and a commercial product, researchers must utilize validated models that satisfy rigorous regulatory submission standards.
Whether you are optimizing a neutralizing antibody or validating a new vaccine development platform, your success depends on the precision of your reagents and the reliability of your models.
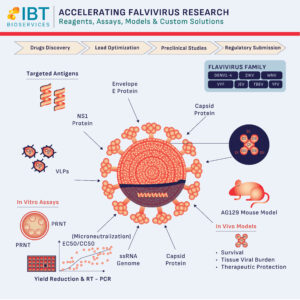
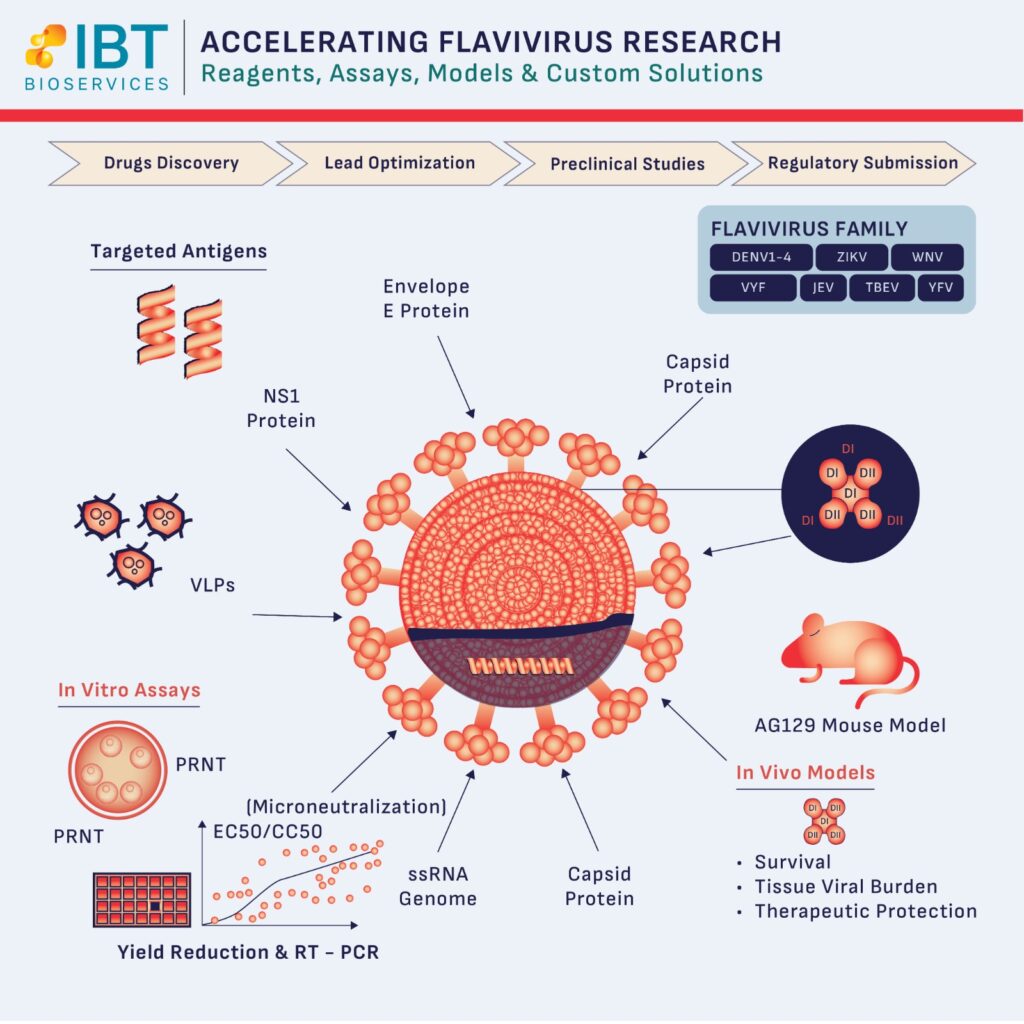
The first hurdle in flavivirus research is often the availability of high-quality reagents. Research is only as good as the antigens used. By utilizing a wide array of virus particles, virus-like particles (VLPs), and recombinant proteins—such as NS1 and Envelope (E) proteins—researchers can better understand the antibody response and immunogenicity of their candidates.
IBT Bioservices supports this foundational stage by providing specialized reagents that target diverse antigens across the flavivirus family, ensuring your ELISA testing and serology work is both accurate and reproducible.
In the lead optimization phase, speed and precision are paramount. Utilizing in vitro antiviral assays allows for the rapid assessment of a compound’s efficacy.

One of the greatest obstacles in preclinical studies is finding an animal model that accurately mimics human disease without the need for mouse-adapted viral strains.
The AG129 mouse model has emerged as a critical tool for this transition. Because these mice are deficient in IFN alpha-beta and gamma receptors, they are highly susceptible to non-adapted flaviviruses, allowing researchers to study:

No two antiviral drug development programs are identical. Sometimes, “off-the-shelf” isn’t enough. Custom assay development allows researchers to adapt platforms to emerging strains or novel mechanisms of action, providing the robust data necessary for a successful regulatory submission.
The financial engine driving flavivirus research is heavily centralized. Global investment in neglected and emerging infectious diseases (EIDs) is estimated in the billions, but the distribution of these funds highlights a significant reliance on American infrastructure.
From benchtop screening to sophisticated in vivo modeling, having an integrated partner can significantly reduce the time to market for antiviral therapeutics.
Ready to move your flavivirus research forward? Contact IBT Bioservices today to learn how our specialized reagents and virology CRO expertise can provide the critical data you need for your next breakthrough.
Leverage our scientific expertise to design a high-impact study for your project.
Our existing partners love boasting about our collaborations! We are happy to provide referrals if needed.
Subscribe to the IBT Bioservices Tech Talk list and receive periodic updates on industry news and IBT Bioservices products and services.