

IBT specializes in delivering top-notch reagents tailored for research and development purposes.
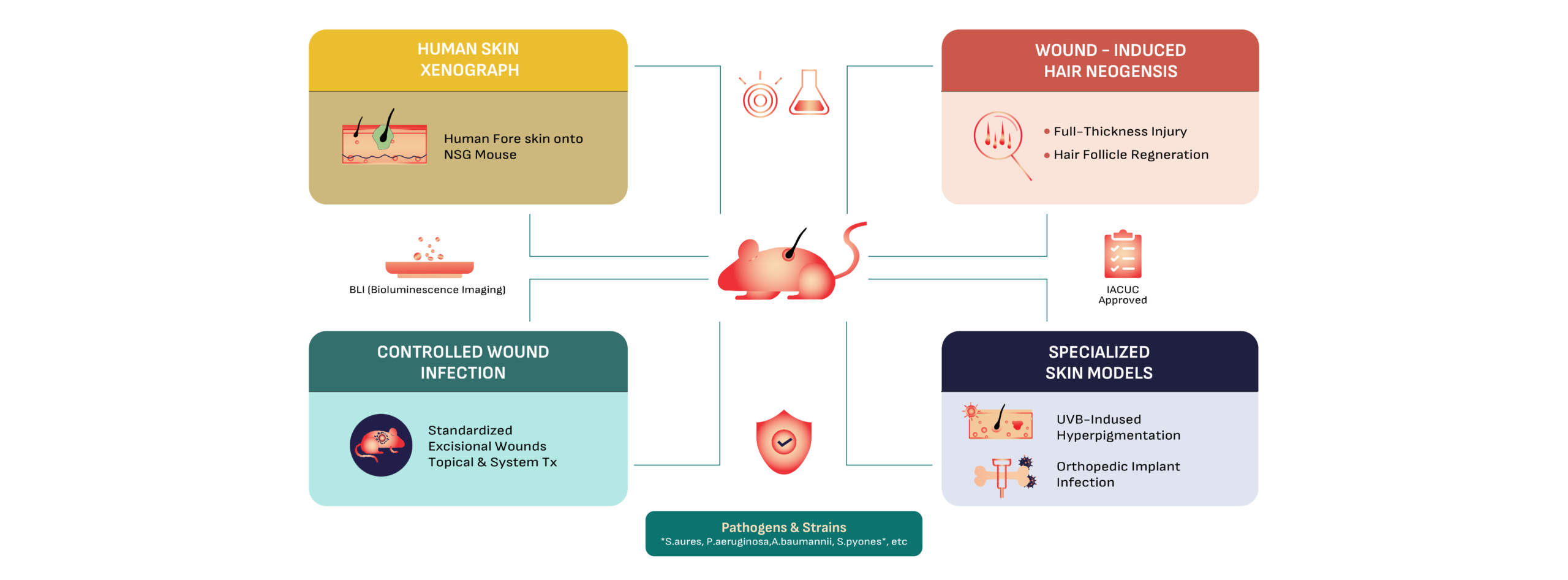
IBT BioServices provides a robust platform of specialized preclinical dermatology models designed to answer critical efficacy and safety questions for your drug development program.
From evaluating the potency of novel topicals against multi-drug resistant pathogens like S. aureus, P. aeruginosa, A. baumannii to observing complex tissue regeneration in our hair neogenesis models, we provide the data-driven insights needed to move candidates into the clinic.
Antimicrobial Efficacy: Does your candidate significantly reduce bacterial burden in colonized wounds or deep-tissue infections?
Drug Delivery & Bioavailability: Is your topical formulation reaching the dermis at therapeutic concentrations, even in the presence of biofilms or scabs?
Inflammation & Pigmentation:Does your treatment mitigate chronic UV-induced damage or hyperpigmentation?
Human Translation: How does your compound interact with human skin tissue in a living, vascularized system?
The Model: Incisional /Excisional wounds over 6-8 days
Capabilities: Evaluate topical vs. systemic (IP) delivery; Wound Area; standardized back titration; and longitudinal tracking of infection via BLI.
Pathogens Supported: Staphylococcus aureus, Pseudomonas aeruginosa, Acinetobacter baumannii AB5075 & more.

The Model: This model assesses the regeneration of hair follicles following a standardized full-thickness injury.
Key Endpoints: Quantitative follicle counting via confocal scanning laser microscopy and wound closure tracking (ImageJ analysis).
Infection Integration: Pathogens can be introduced beneath the scab on Day 3 to evaluate the impact of infection on tissue regeneration.

The Model: Chronic, low-dose UVB exposure over 28 days.
Readouts: Pigmentation scoring, ear thickness, and detailed histopathology to assess skin barrier damage and recovery.

The Model: Human foreskin is grafted onto immunocompromised (NSG) mice, creating a chimeric system with living human skin.
Why Use It: It is the “gold standard” for translational research, allowing for the study of human-specific skin responses and pathogens.
Pathogens Supported: P. aeruginosa, S. pyogenes, S. aureus, and S. epidermidis.
Timeline: 7–28 days.

High-sensitivity Bioluminescence Imaging (BLI) for non-invasive, longitudinal tracking of bacterial burden.

Standardized back titration and CFU enumeration from tissue, sutures, and orthopedic implants.

Approved IACUC protocols for most standard infection models. (Note: UVB and Xenograft models require a 1-2 month activation TAT).

Models available in Wildtype (C57BL/6), Diabetic (db/db), Immunocompromised (NSG), or custom knockout strains.
We maintain a validated library of skin-relevant pathogens:
Partner with IBT BioServices for high-sensitivity, reliable preclinical data. Our team is ready to help you customize a study design that fits your specific therapeutic goals.
I have a specific protocol in mind
I need help choosing the right model
I want to see your other bacterial capabilities
I want to see your immunology capabilities