

IBT specializes in delivering top-notch reagents tailored for research and development purposes.
At IBT, we offer Pseudovirus Neutralization Assay Services to assess the ability of antibodies to neutralize the infectivity of a pseudovirus, which are created by packaging a non-replicating viral genome with the envelope proteins or surface antigens of another virus. Pseudoviruses are designed for various research purposes, particularly in virology and immunology, to study the infectivity and neutralization of specific viral proteins or antigens.
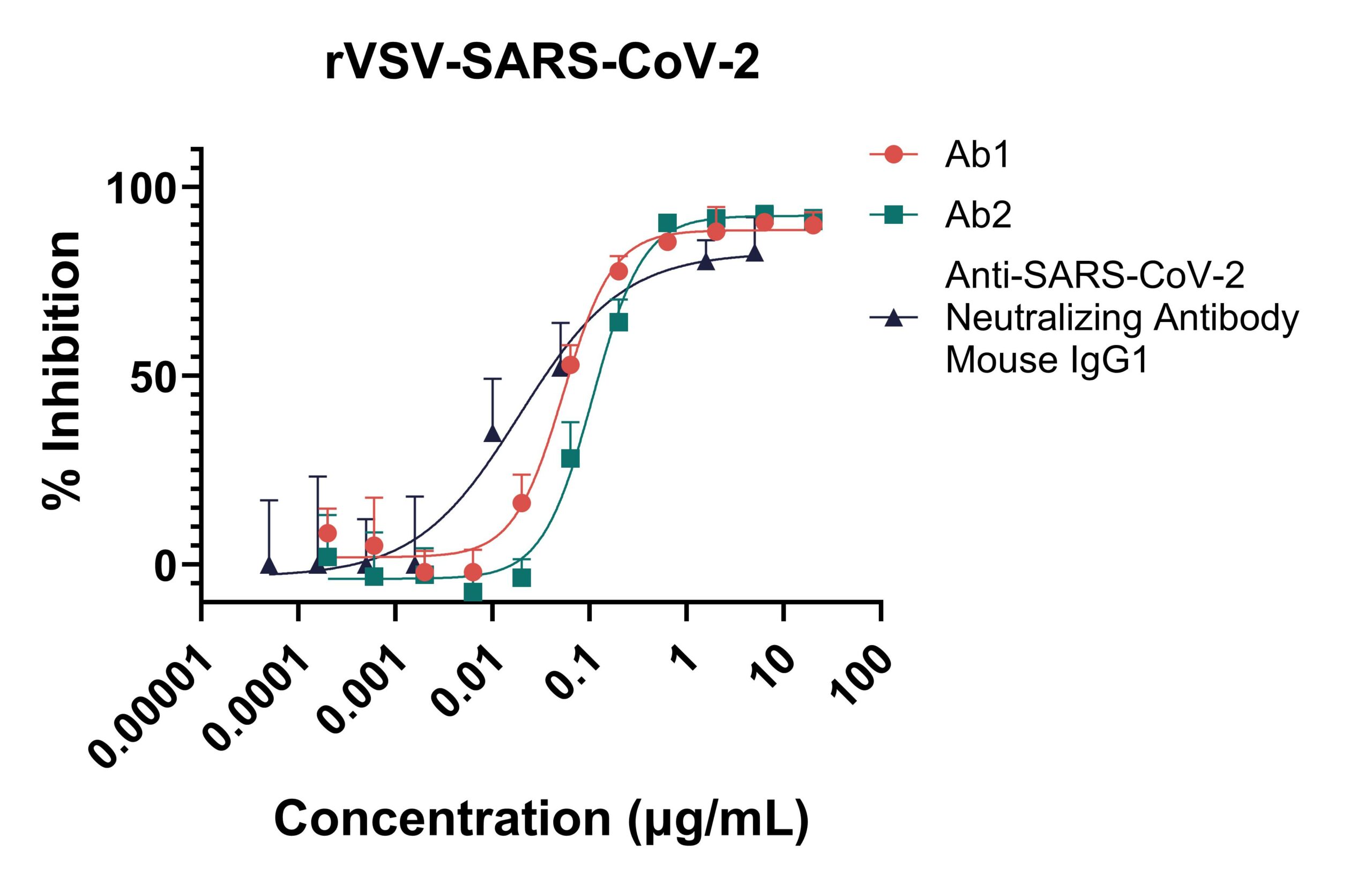
Ab1 and Ab2 are antibodies tested against pseudovirus rVSV-SARS-CoV-2 (Wuhan strain) in our Pseudovirus NeutralizationAssay (PsVNA). Both antibodies exhibit similar neutralization profiles and have comparable neutralizing activity. A commercially available IgG1 anti-SARS-CoV-2 neutralizing antibody is included as a positive control.
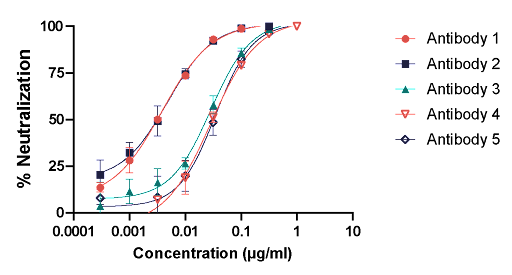
Antibodies 1-5 were assessed for the neutralization capability against rVSV-MARV Angola in a pseudovirus neutralization assay. All antibodies demonstrate neutralization of rVSV-MARV Angola. The neutralization curves for Antibodies 1 and 2 have similar profiles and more neutralizing potency is observed in Antibodies 1 and 2 when compared to Antibodies 3, 4, and 5.
The pseudovirus neutralization assay is a valuable methodology in virology and immunology for studying the neutralizing capacity of antibodies against specific viral proteins or antigens. The pseudovirus neutralization assay is based on the principle that neutralizing antibodies have the capacity to block the infectivity of pseudoviruses, which express the surface proteins of the target virus. This assay allows the study of specific antibodies that target viral entry mechanisms.
With years of experience in the field, our team of skilled scientists and technicians are well-versed in conducting assays involving a wide range of pseudoviruses. We utilize state-of-the-art equipment and follow stringent quality control measures to ensure precise and reproducible results.
Pseudovirus Neutralization Assays are essential for assessing the efficacy of vaccines by measuring their ability to induce neutralizing antibodies against specific viral proteins or antigens.
These assays allow the study of antibodies targeting the viral entry process, a critical step in the viral life cycle.
Pseudovirus Neutralization Assays are widely used in the investigation of the immune response to viral infections and the evaluation of antiviral treatments.
Researchers can use pseudoviruses carrying different viral variants or mutants to study how neutralizing antibodies respond to various strains of the virus.

Our team of scientists and technicians are highly skilled and experienced in conducting HAI assays, ensuring accurate and reliable results.

We understand the unique needs of each research project and provide customized assay designs to meet your specific requirements.

We prioritize efficiency without compromising on quality. Our streamlined processes and advanced equipment allow for quick turnaround times, ensuring you receive results promptly.

We maintain rigorous quality control measures throughout our operations, ensuring the accuracy and reproducibility of our assay results.

Our dedicated customer support team is always available to address your queries and provide assistance at every step of the process.