

IBT specializes in delivering top-notch reagents tailored for research and development purposes.
A viral yield reduction assay is used to evaluate the effectiveness of antiviral agents, drugs, or experimental treatments in reducing the total production of virus particles by infected host cells. This assay is essential in virology and drug development for assessing the impact of potential treatments on viral replication and productivity
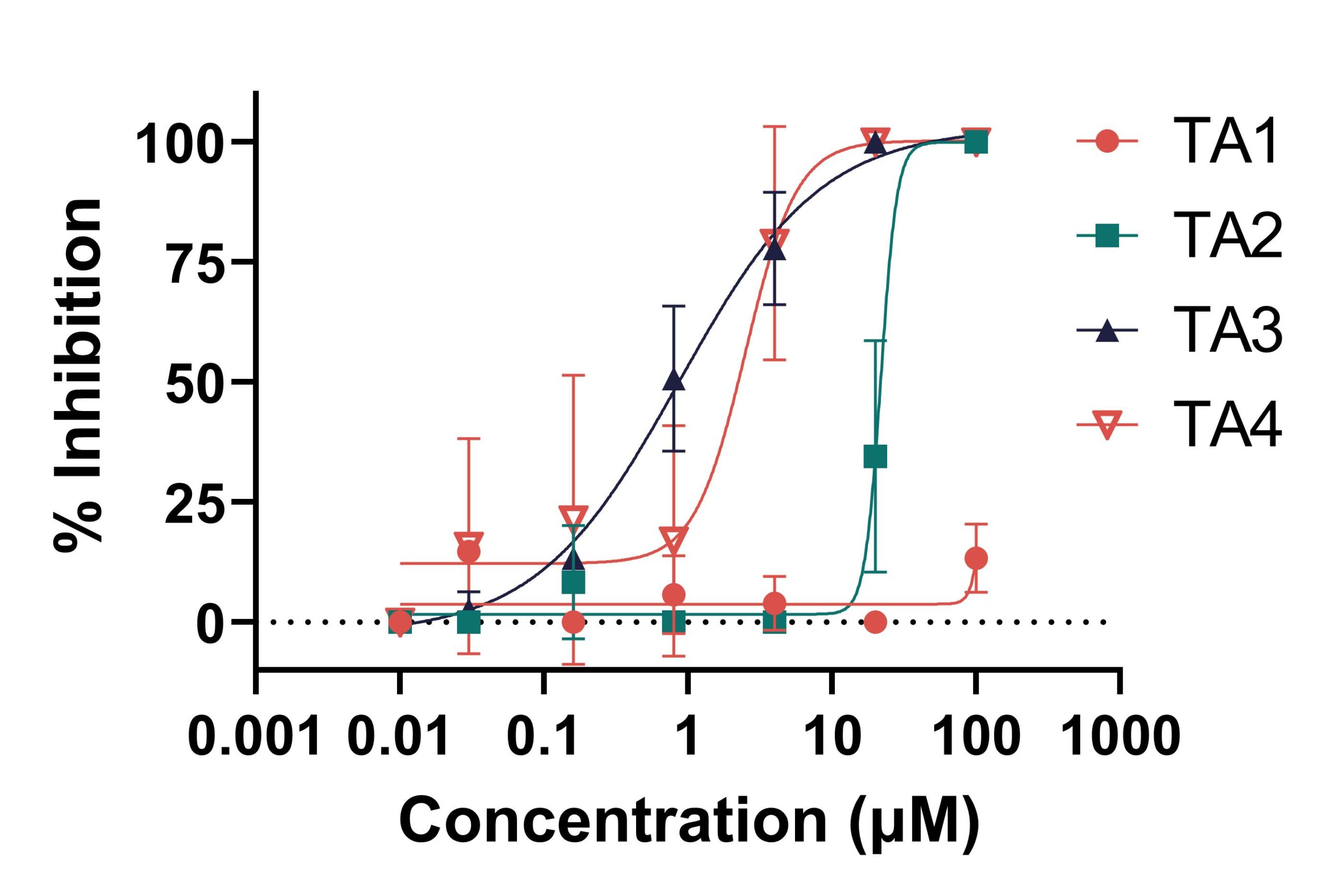
Test articles (TAs)1-4 are products tested in a yield reduction assay against IFV-A/PR/8/34 in MDCK cells. Among all tested compounds, the largest degree of % inhibition was observed in cells treated with TA3. Conversely, the lowest antiviral efficacy against IFV-A/PR8 is observed in cells treated with TA2.
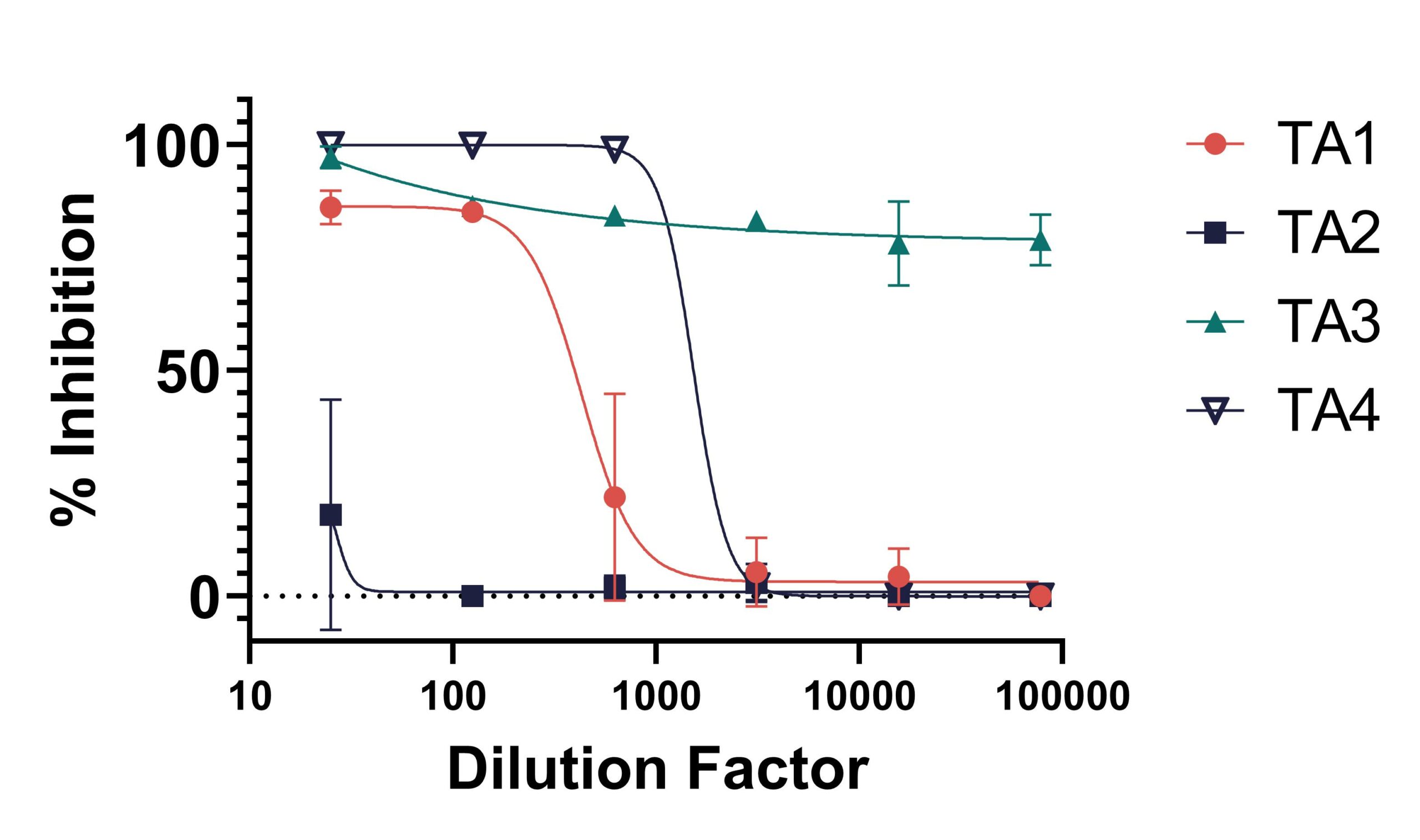
Test articles (TAs) 1-4 are compounds that were tested for antiviral efficacy against DENV-2 NGC in a yield reduction assay in Vero cells. The compounds were incubated with the cells for a predetermined amount of time with a known amount of DENV-2 NGC. Supernatants were collected and the amount of virus in each sample was quantified via plaque assay. % inhibition was calculated by comparing the reduction of viral titer when compared to the virus only control.
The yield reduction assay is based on the principle that antiviral agents, when effective, reduce the overall yield of virus particles produced by infected cells. By measuring the reduction in viral production in the presence of these agents, researchers can assess their antiviral efficacy.
Yield reduction assays are crucial methodologies for assessing the antiviral efficacy of drugs and treatments, contributing significantly to the development of antiviral therapies and the understanding of viral diseases. They provide important insights into the impact of various treatments on viral replication and productivity.
With years of experience in the field, our team of skilled scientists and technicians are well-versed in conducting yield reduction assays for a wide range of pathogens, including influenza and dengue viruses. We utilize state-of-the-art equipment and follow stringent quality control measures to ensure precise and reproducible results.
Viral yield reduction assays are vital for evaluating the effectiveness of potential antiviral drugs in reducing viral replication and productivity.
Viral yield reduction assays can be used to study the development of drug resistance in viruses by exposing them to antiviral agents over time.
Researchers use yield reduction assay to establish the minimum effective concentration of an antiviral compound required to inhibit viral replication.
Assessing the reduction in viral yield in the presence of vaccines helps determine their potential to reduce or prevent viral infections.

Our team of scientists and technicians are highly skilled and experienced in conducting HAI assays, ensuring accurate and reliable results.

We understand the unique needs of each research project and provide customized assay designs to meet your specific requirements.

We prioritize efficiency without compromising on quality. Our streamlined processes and advanced equipment allow for quick turnaround times, ensuring you receive results promptly.

We maintain rigorous quality control measures throughout our operations, ensuring the accuracy and reproducibility of our assay results.

Our dedicated customer support team is always available to address your queries and provide assistance at every step of the process.